 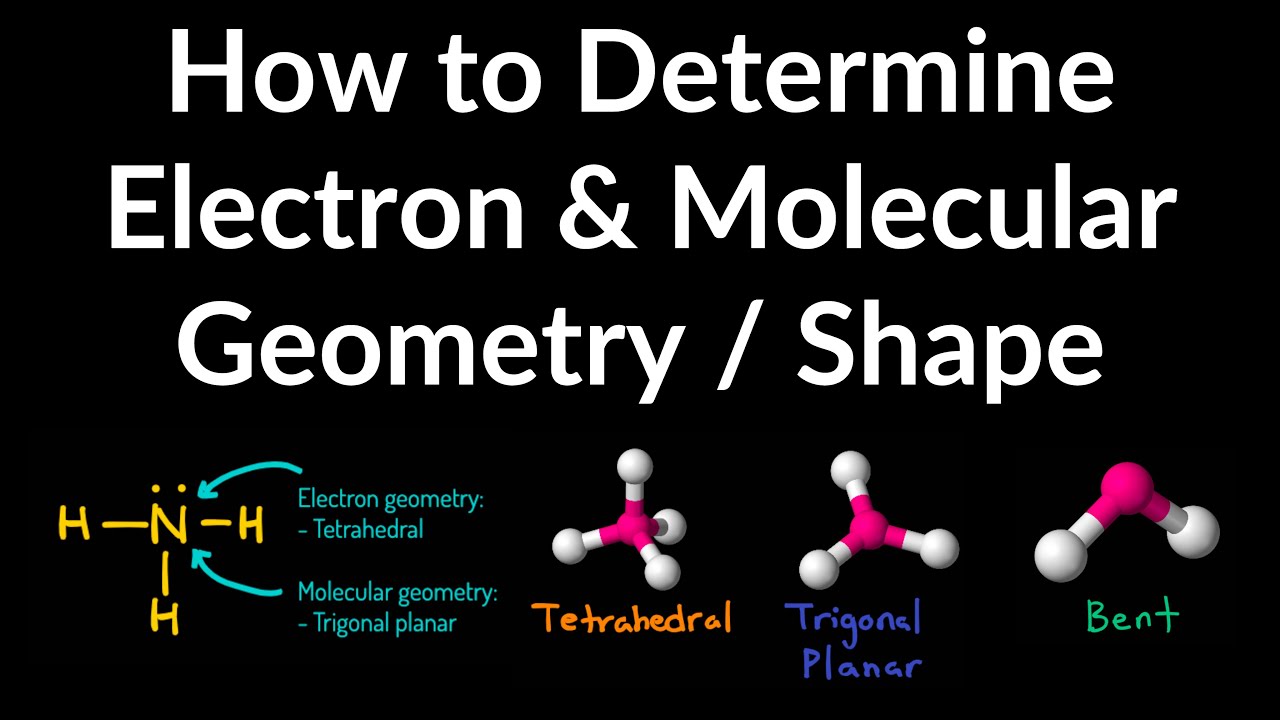 
A comma delimited list of several species may be entered. Therefore, the molecular geometry for BH 3 is trigonal planar and its electron geometry is also trigonal planar. This means that CH3 (CH2)4CH3 will be treated the same as C6H14. As per the VSEPR chart, if a molecule gets AX 3 generic formula then its molecular geometry will be trigonal planar and electron geometry will also be trigonal planar. Electron geometry helps us in determining the arrangement of various electron groups. This gives a molecule a specific shape and bond angles. When the two electron groups are 180° apart, the atoms attached to those electron groups are also 180° apart, so the overall molecular shape is linear. Multiple specifications for an atom will be added. Electron geometry and molecular geometry are the arrangements of electrons or atoms in three-dimensional space around a central atom. CH4 molecular geometry is tetrahedral and its electron geometry is also tetrahedral. Electron domain geometry vs molecular geometry Electron domain Molecular geometry: definition, chart, shapes, and examplesElectron bonding covalent geometrics geometries ib overlap orbitals sigma. A molecule whose central atom contains only two electron groups orients those two groups as far apart from each other as possible-180° apart. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. Remember that a multiple bond counts as only one electron group.Īny molecule with only two atoms is linear. Molecular shapes geometry electron domain chemistry chart pair geometries pairs shape molecules four two three atom central oh atoms google Electron pair geometry and molecular geometry chart Chem college: electron geometry and steric number. When applying VSEPR to simple molecules, the first thing to do is to count the number of electron groups around the central atom. There are two types of electron groups A covalent bond of any type or a lone electron pair.: any type of bond-single, double, or triple-and lone electron pairs. VSEPR makes a distinction between electron group geometry, which expresses how electron groups (bonds and nonbonding electron pairs) are arranged, and molecular geometry, which expresses how the atoms in a molecule are arranged. (a) BH3 There are 3 valence electrons in boron, and 1 each in hydrogen, so there are 6 electrons total. They help in predicting molecular behavior, reactivity, and the physical properties of compounds. Solution: First, we draw the Lewis diagram for each structure, then we determine the electron domain geometry, and from there, the molecular geometry. Both Electron Geometry and Molecular Geometry are vital concepts in chemistry, particularly in the understanding of molecular shape and polarity. It basically says that electron pairs, being composed of negatively charged particles, repel each other to get as far away from each other as possible. Example 1: Determine the molecular geometry of: (a) BH3 (b) CH2O (c) NH3 (d) BrF5. Interactive: Electron Geometry: Molecules assume different shapes due to patterns of shared and unshared electrons. The basic idea in molecular shapes is called valence shell electron pair repulsion (VSEPR) The general concept that estimates the shape of a simple molecule. Small molecules-molecules with a single central atom-have shapes that can be easily predicted. There is an abundance of experimental evidence to that effect-from their physical properties to their chemical reactivity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
